Proenkephalin A 119-159
(penKid)
The kidney function biomarker
The current biomarkers used in the critical care routine to detect acute kidney injury (AKI) include serum creatinine and urine output. The inherent limitations of these biomarkers such as delayed detection of kidney deterioration and error proneness, urge the need for novel biomarkers. An emerging body of evidence demonstrates that the biomarker Proenkephalin A 119-159 (penKid*) overcomes these limitations.
Current blind spots of routinely used biomarkers:
- AKI is mostly symptom-free in the early disease stages.
- Current biomarkers show delayed recognition when 50% of kidney function is already lost.
- Standard biomarkers require observation at least at two independent time points.
- Comorbidities, personal factors (e.g. age and muscle mass) and treatments influence currently used biomarkers.
Potential advantages of the kidney function biomarker penKid:
- Showing kidney function in real-time (1).
- Predicting the incidence of AKI within 48h in septic patients (2,3).
- Correlating with the in vivo measurement of glomerular filtration rate (GFR) (4,5,6).
- Reflecting kidney function in adults (7,8,9) and children, representing a biomarker for pediatric AKI (10).
- Independent of inflammation (1,2,3,11) and common comorbidities, e.g. diabetes (2).
The biomarker penKid is convincing in clinical reseach
True GFR representation
PenKid is a blood-based parameter that strongly correlates with true GFR (5,7).
Kidney function in real-time
Monitoring penKid identification of patients with developing AKI (4,5,6,12).
Monitoring patient severity
Relative changes in penKid blood concentration show the improvement or deterioration of kidney function (3,5,6,8).
Applicable in critical conditions
PenKid is independent of inflammation (1,2,3,11) or common comorbidities, e.g. diabetes (2).
scientific evidence
A small molecule becomes a messenger for a serious disease
The best indicator of kidney function is the glomerular filtration rate (GFR) (5,7). However, the gold standard technique for determining the actual GFR relies on complex, time-consuming, expensive intervention using injected tracer substances such as inulin or iothalamate, which is not feasible in daily practice (13). Clinical routine relies on determining an estimated GFR (eGFR) using formulas which have been derived from patients with chronic and stable conditions (14). However, a plethora of non-renal factors (muscle mass, inflammation, among others) influence serum creatinine and thereby the result.
An emerging body of evidence demonstrates that monitoring penKid levels, which is exclusively eliminated through the kidney (43) and thereby reflects the true GFR, overcomes these limitations (4,5,6). PenKid levels reveal kidney function in real-time and offer a blood-based alternative for the complex and time-consuming in vivo measurement of true GFR.
Healthy state
Kidney function is stimulated by the hormone Enkephalin (15) which itself is difficult to detect due to its instability. During enkephalin processing, the stable fragment Proenkephalin A 119-159 (penKid) is generated. Quantification of penKid overcomes this analytical limitation and enables indirect assessment of kidney function.
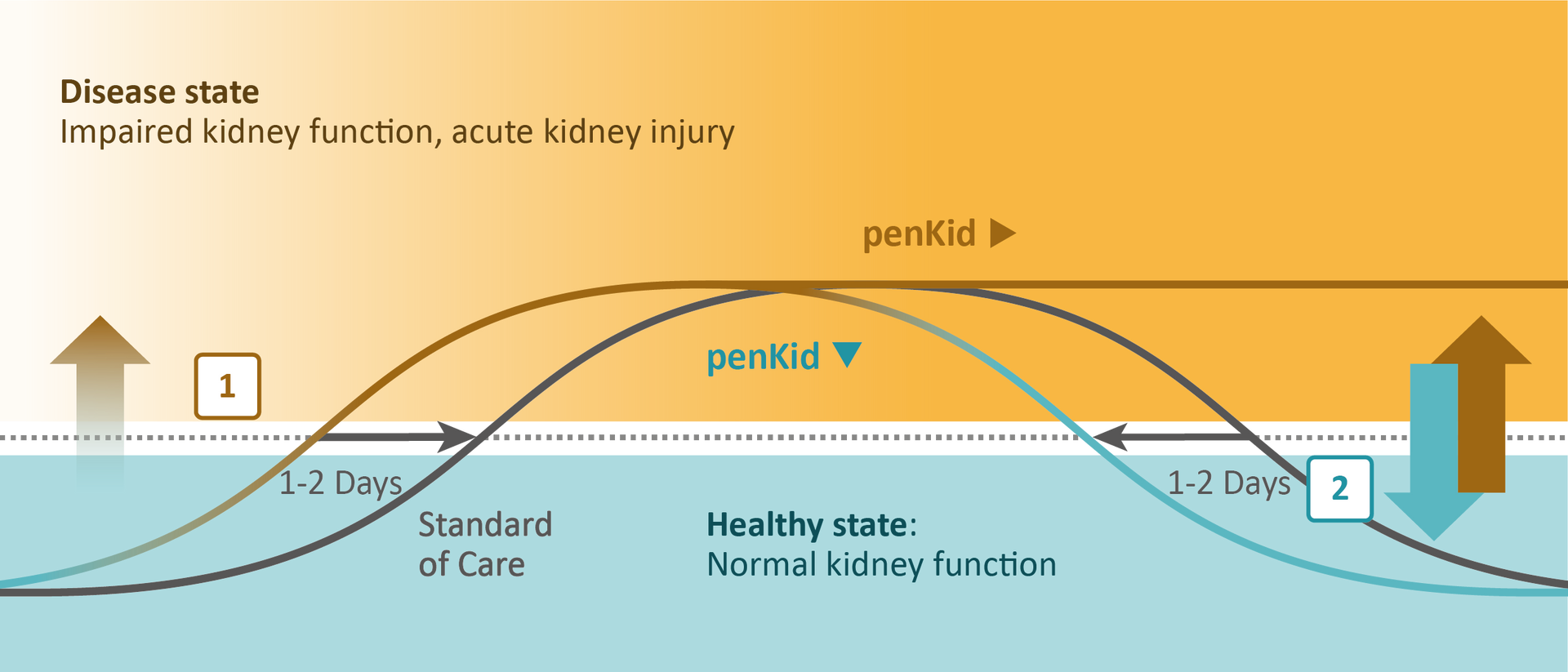
Disease state
The administration of nephrotoxic drugs or critical illness can lead to an impairment of kidney function, which may increase enkephalin production to stimulate renal function, by increasing urine flow and sodium excretion (15). The kidney status can thus be indirectly assessed by measuring penKid blood levels, which reflects kidney function in real-time and remains independent of inflammation (1,2,3,11) and common comorbidities (2).
Monitoring kidney function in real-time using the biomarker penKid
SCIENTIFIC EVIDENCE
Independent of common comorbidities (e.g. diabetes) and the frequently occurring systemic inflammation in critically ill patients, rising penKid blood levels [1] predict acute kidney injury up to 48 hours earlier than today’s standard of care (16). Importantly, decreasing penKid blood levels [2] as a consequence of early resuscitation indicates the improvement of kidney function (2).
| Pooled sensitivity | 0.69 (95% CI 0.62-0.75) |
|---|---|
| Pooled specificity | 0.76 (95% CI 0.68-0.82) |
| Positive likelihood ratio | 2.83 (95% CI 2.06-3.88) |
| Negative likelihood ratio | 0.41 (95% CI 0.33-0.52) |
| Area under the curve | 0.77 (95% CI 0.73-0.81) |
Table 1: Pooled performance criteria of penKid for early detection of AKI. Results from a systematic review with meta-analysis incorporating 11 observational studies with 3,969 patients and 23.4% AKI incidence (33).

Clinical research
Sepsis and septic shock
Intensive care units (ICU): Critically ill patients with sepsis or septic shock admitted to the ICU require efficient diagnostic strategies to predict acute kidney deterioration and enable monitoring of recovery of renal function. PenKid predicted major adverse kidney events, transient AKI, worsening of renal function, and renal recovery in ICU patients (2,3).
Clinical performance characteristics: 529 adult patients with sepsis or septic shock and available AKI diagnoses were enrolled in a clinical performance study (34). Performance was calculated for identification of patients with an increase of serum creatinine (in respect to admission baseline values) within 48 hours after enrolment in the study.
Heart failure
In heart failure, elevated penKid levels provide important prognostic information across the disease spectrum. In acute heart failure (AHF), particularly in patients with renal dysfunction and inadequate decongestion during hospitalization, a rapid increase in penKid helps identify individuals who do not tolerate intensified diuretic therapy and predicts worsening renal function, rehospitalization, in-hospital and long-term mortality (9,18,19,20, 35, 36). In chronic heart failure (CHF), especially in heart failure with preserved ejection fraction (HFpEF), where effective monitoring biomarkers are lacking, high penKid levels have been associated with heart failure rehospitalization (21) and recurrence of acute myocardial infarction (22). Furthermore, in stable ambulatory patients with heart failure, increased penKid levels demonstrate significant predictive value for risk stratification (23), supporting early identification of patients at risk of organ failure and poor outcomes.
Kidney transplantation
PenKid is associated with kidney function in renal transplant recipients (RTR) and in healthy kidney donors (27). A common complication after kidney transplantation is delayed graft function (DGF) (37). PenKid provides a significant clinical advantage by identifying patients at risk for DGF well before traditional biomarkers like serum creatinine show any changes (38). Moreover, penKid distinguishes between slow- and delayed graft function with remarkable accuracy, up to eight days earlier than conventional assessment (38). Furthermore, increased penKid levels are associated with an elevated risk of long-term graft failure (27).
Renal replacement therapy
Critically ill patients are often at risk of developing AKI and are likely in need of organ support, such as kidney replacement therapy (KRT) (30). The monitoring of kidney function under KRT is crucial to guide successful discontinuation of therapy. However, current clinical practice, which– besides clinical characteristics – heavily relies on urine output, requires improvement for an informed decision-making to discontinue KRT. Plasma concentrations of penKid are maintained under KRT (39), making penKid a valuable tool for monitoring patients under KRT and for guiding KRT discontinuation. Recent data has shown that penKid levels at CKRT discontinuation discriminate between patients successfully and unsuccessfully liberated from CKRT (40).
Pediatric ICU
Pediatric AKI is often diagnosed too late for successful therapeutic interventions, leading to adverse outcomes and chronic kidney impairment (41). PenKid identifies AKI and its severity in critically ill neonates and children under one year of age, offering a potential solution to the shortcomings in pediatric critical care (10,42).
PenKid Whitepaper
The whitepaper covers practical insights on the AKI diagnostics and highlights the scientific evidence for penKid, a kidney function biomarker.
*Disclaimer
ELISA sphingotest® penKid®, sphingotest® penKid®, and sphingotest® bio-ADM® are offered for research use only. “penKid” and “bio-ADM” represent the analytes Proenkephalin A 119-159 and bioactive Adrenomedullin 1-52, respectively.
Reference Literature
(1) Marino et al. (2015), Diagnostic and short-term prognostic utility of plasma proenkephalin (pro-ENK) for acute kidney injury in patients admitted with sepsis in the emergency department, J Nephrol,
View the paper
(2) Hollinger et al. (2018), Proenkephalin A 119-159 (Penkid) is an early biomarker of septic acute kidney injury - the kidney in sepsis and septic shock (Kid-SSS) study, Kidney Int Rep.
View the paper
(3) Caironi et al. (2018), Circulating Proenkephalin, Acute Kidney Injury, and Its Improvement in Patients with Severe Sepsis or Shock. Clin Chem.
View the paper
(4) Beunders et al. (2017), Proenkephalin (PENK) as a novel biomarker for kidney function, JALM.
View the paper
(5) Beunders et al. (2020), Proenkephalin compared to conventional methods to assess kidney function in critically ill sepsis patients, Shock.
View the paper
(6) Khorashadi et al. (2020), Proenkephalin: A New Biomarker for Glomerular Filtration Rate and Acute Kidney Injury. Nephron.
View the paper
(7) Donato et al. (2018), Analytical performance of an immunoassay to measure proenkephalin. Clin Biochem.
View the paper
(8) Schulz et al. (2017), High Level of Fasting Plasma Proenkephalin-A Predicts Deterioration of Kidney Function and Incidence of CKD. J Am Soc Nephrol.
View the paper
(9) Emmens et al. (2019), Proenkephalin, an Opioid System Surrogate, as a Novel Comprehensive Renal Marker in Heart Failure. Circ Heart Fail.
View the paper
(10) Hartman et al. (2020), Proenkephalin as a new biomarker for pediatric acute kidney injury - reference values and performance in children under one year of age. Clin Chem Lab Med.
View the paper
(11) Kim et al. (2017), Proenkephalin, Neutrophil Gelatinase-Associated Lipocalin, and Estimated Glomerular Filtration Rates in Patients With Sepsis. Ann Lab Med.
View the paper
(12) Dépret et al. (2020), PenKid measurement at admission is associated with outcome in severely ill burn patients. Burns.
View the paper
(13) Bjornstad et al. (2018), Measured GFR in Routine Clinical Practice-The Promise of Dried Blood Spots. Adv Chronic Kidney Dis.
View the paper
(14) Schaeffner (2017), Determining the Glomerular Filtration Rate-An Overview. J Ren Nutr.
View the paper
(15) Sezen et al. (1998), Renal Excretory Responses Produced by theDelta Opioid Agonist, BW373U86, in Conscious Rats. Journal of Pharmacology and Experimental Therapeutics.
View the paper
(16) Lima et al. (2022), Role of proenkephalin in the diagnosis of severe and subclinical acute kidney injury during the perioperative period of liver transplantation. Pract Lab Med.
View the paper
(17) Rosenqvist et al. (2019), Proenkephalin a 119-159 (penKid) - a novel biomarker for acute kidney injury in sepsis: an observational study. BMC Emerg Med.
View the paper
(18) Molvin et al. (2019), Bioactive adrenomedullin, proenkephalin A and clinical outcomes in an acute heart failure setting. Open Heart.
View the paper
(19) Ng et al. (2017), Proenkephalin, Renal Dysfunction, and Prognosis in Patients With Acute Heart Failure: A GREAT Network Study. J Am Coll Cardiol.
View the paper
(20) Matsue et al. (2017), Clinical Correlates and Prognostic Value of Proenkephalin in Acute and Chronic Heart Failure. J Card Fail.
View the paper
(21) Kanagala et al. (2019), Proenkephalin and prognosis in heart failure with preserved ejection fraction: a GREAT network study. Clin Res Cardiol.
View the paper
(22) Ng et al. (2014), Proenkephalin and prognosis after acute myocardial infarction. J Am Coll Cardiol.
View the paper
(23) Arbit et al. (2016), Prognostic Usefulness of Proenkephalin in Stable Ambulatory Patients With Heart Failure. Am J Cardiol.
View the paper
(24) Shah et al. (2015), Proenkephalin predicts acute kidney injury in cardiac surgery patients. Clin Nephrol.
View the paper
(25) Mossanen et al. (2017), Elevated Soluble Urokinase Plasminogen Activator Receptor and Proenkephalin Serum Levels Predict the Development of Acute Kidney Injury after Cardiac Surgery. International Journal of Molecular Sciences.
View the paper
(26) Gombert et al. (2020), Proenkephalin A 119-159 May Predict Post-operative Acute Kidney Injury and in Hospital Mortality Following Open or Endovascular Thoraco-abdominal Aortic Repair. Eur J Vasc Endovasc Surg.
View the paper
(27) Kieneker et al. (2017), Plasma Proenkephalin and Poor Long-Term Outcome in Renal Transplant Recipients. Transplant Direct.
View the paper
(28) Kim et al. (2020), Proenkephalin Predicts Organ Failure, Renal Replacement Therapy, and Mortality in Patients with Sepsis. Ann Lab Med.
View the paper
(29) Andreoli (2009), Acute kidney injury in children. Pediatr Nephrol.
View the paper
(30) Ali et al. (2018), The Prevention and Management of Contrast-induced Acute Kidney Injury: A Mini-review of the Literature. Cureus.
View the paper
(31) von Groote et al. (2022), Proenkephalin A 119-159 predicts early and successful liberation from renal replacement therapy in critically ill patients with acute kidney injury: a post hoc analysis of the ELAIN trial. Crit Care.
View the paper
(32) von Groote et al. (2023), Evaluation of Proenkephalin A 119-159 for liberation from renal replacement therapy: an external, multicenter pilot study in critically ill patients with acute kidney injury. Crit Care.
View the paper
(33) Lin LC et al. (2023) Proenkephalin as a biomarker correlates with acute kidney injury: a systematic review with meta-analysis and trial sequential analysis. Critical care.
(34) Schulte J et al. (2024), Clinical performance of proenkephalin A 119-159 for the early diagnosis of acute kidney injury in patients with sepsis or septic shock. medRxiv preprint.
(35) Siranart N et al. (2023) Proenkephalin as a Novel Prognostic Marker in Heart Failure Patients: A Systematic Review and Meta-Analysis. Int J Mol Sci.
(36) Matsiras D et al. (2025) Predictive Value of Point-of-Care Proenkephalin for Worsening Renal Function and Mortality in Patients Presenting to Emergency Department with Acute Heart Failure. J Clin Med.
(37) Yarlagadda SG et al. (2009) Association between delayed graft function and allograft and patient survival: a systematic review and meta-analysis. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association.
(38) Benning L et al. (2025) Proenkephalin A 119-159 in Kidney Transplantation: A Novel Biomarker for Superior Tracking of Graft Function Trajectories. Transpl Int.
(39) Martin L et al. (2025) Implementation and One-Year Evaluation of Proenkephalin A in Critical Care. International journal of molecular sciences.
(40) Tichy J et al. (2024) Prediction of Successful Liberation from Continuous Renal Replacement Therapy Using a Novel Biomarker in Patients with Acute Kidney Injury after Cardiac Surgery-An Observational Trial. International journal of molecular sciences.
(41) Lebel A et al. (2020) Long-term complications of acute kidney injury in children. Curr Opin Pediatr.
(42) Smeets NJL (2022) Proenkephalin A as a marker for glomerular filtration rate in critically ill children: validation against gold standard iohexol GFR measurements. Clinical chemistry and laboratory medicine: CCLM / FESCC.
(43) Sigurjonsson J et al. (2025) Renal elimination and eGFR prediction of proenkephalin. Scand J Clin Lab Invest.