SphingoTec's biomarker penKid® shows best representation of true glomerular filtration rate and has utility in patients with sever burns two studies show
- In-depth method comparison by Dutch group shows that sphingotec's proprietary kidney function biomarker penKid® is currently the most accurate surrogate marker for true glomerular filtration rate in patients with renal impairment
- Data by PRONOBURNS group show that penKid® accurately predicts acute kidney injury (AKI) in patients with burns, adding benefit on top of standard testing
- Since January 2020, an automated CE-marked IVD penKid® assay developed for sphingotec's proprietary point-of-care platform Nexus IB10 is available for use in critical care settings
Hennigsdorf/Berlin, Germany, February 19, 2020
- Diagnostics company SphingoTec GmbH ("sphingotec", Hennigsdorf Germany) today announced the publication of two studies demonstrating that its kidney function biomarker Proenkephalin (penKid®) is the most accurate surrogate for assessing true glomerular filtration rate (true GFR) and is reliably predictive for acute kidney injury (AKI) in patients with severe burns. Although AKI is a major complication in critically ill patients, current diagnostic standard methods do not properly and timely diagnose impaired renal function. The two recently published studies add to the rapidly growing body of evidence suggesting that penKid® can address this highly unmet diagnostic need.
In Shock[1], a team headed by Prof. Peter Pickkers from Radboud UMC (Nijmegen, The Netherlands) confirmed by in-depth diagnostic method comparison in patients with impaired kidney function that penKid® levels properly reflect kidney function. While today’s standard of care uses estimations of the glomerular filtration rate (eGFR) to assess renal impairment, the current study shows that these methods overestimate true GFR with over 30%. The published findings demonstrate that penKid® can add value by properly reflecting true GFR that can otherwise only be measured using in vivo clearance of iohexol, an invasive method too laborious and time-consuming for clinical routine use.
Another study published in the Journal of Burns[2] reports for the first time that high penKid® plasma levels at admission to the intensive care unit (ICU) were associated with the risk for developing AKI in patients with severe burns, where mortality rates range from 30-70%.The new data suggest, that the current renal function- and AKI standard markers should be complemented with penKid® values for accurately quantifying the kidney function in critically ill patients.
Dr. Andreas Bergmann, CEO of sphingotec, commented: “penKid® is an early renal function biomarker that is not biased by co-morbidities while reflecting true GFR. penKid® has been tested for the first time to identify ICU patients with severe burns that need rapid and aggressive intervention to prevent mortality caused by AKI.”
To support timely treatment decisions that are likely to improve patient management in critical care patients, sphingotec launched a fully automated CE-IVD-marked penKid® assay on its Nexus IB10 platform in January 2020. This new test complements a wide-range of assays for acute care settings that are already available on this widely used point-of-care platform that can be flexibly deployed in near-patient as well as laboratory settings.
###
References
1. Beunders, R. et al., Proenkephalin compared to conventional methods to assess kidney function in critically ill sepsis patients, Shock,, doi:10.1097/SHK.0000000000001510
2. Depret, F. et al, PenKid measurement at admission is associated with outcome in severely ill burn patients, J.Burns, doi: doi.org/10.1016/j.burns.2020.01.002
About sphingotec
SphingoTec GmbH ("sphingotec"; Hennigsdorf near Berlin, Germany) develops and markets innovative in vitro diagnostic (IVD) tests for novel and proprietary biomarkers for the diagnosis, prediction and monitoring of acute medical conditions, such as sepsis, acute heart failure, circulatory shock, and acute kidney injury in order to support patient management and provide guidance for treatment strategies. sphingotec's proprietary biomarker portfolio includes Bioactive Adrenomedullin (bio-ADM®), a unique biomarker for real-time assessment of endothelial function in conditions like sepsis or congestive heart failure, Proenkephalin (penKid®), a unique biomarker for real-time assessment of kidney function, and Dipeptidyl Peptidase 3 (DPP3), a unique biomarker for cardio-renal pathway disruptions leading to acute organ dysfunction. In addition, sphingotec develops a portfolio of novel biomarkers, which predict the risks of developing obesity, breast cancer and cardiovascular diseases. IVD tests for sphingotec’s proprietary biomarkers are made available as sphingotest® microtiterplate tests as well as point-of-care tests on the Nexus IB10 immunoassay platform by sphingotec’s subsidiary Nexus Dx Inc. (San Diego, CA, USA) alongside a broad menu of IB10 tests for established biomarkers for acute and critical care.
About penKid®
sphingotest® penKid® measures proenkephalin (penKid®), a stable fragment of the kidney stimulating hormone enkephalin. penKid® has been demonstrated to be a real-time surrogate biomarker for glomerular filtration rate, the gold standard to assess renal function. Measuring penKid® blood concentrations allows for timely information on kidney function in critically ill patients. Early assessment of worsening and improving of renal function on intensive care units and in emergency departments allows adjustment of nephrotoxic drug administration and the initiation of kidney-protective strategies to prevent acute kidney injury and thereby improve outcomes.
About Nexus Dx Inc. and the IB10 Platform
Nexus Dx Inc., a wholly-owned subsidiary of sphingotec, headquartered in San Diego, CA, USA, is a global provider of a near patient testing system and advanced diagnostic solution. The company is improving patient care by providing the medical community with rapid and reliable information at the point of care (POC), delivering patient information when and where it is needed most. The company has invested over $160m to develop and market the IB10 analyzer system which, without the need for sample preparation, automatically separates plasma from whole blood with subsequent reliable and quantitative detection of biomarkers in the plasma by means of antibodies. With a hands-on-time of less than 3 minutes the easy-to-use system provides in only 20 minutes test results for biomarkers that are crucial in the management of critical care patients. The portfolio of IB10 assays includes tests for established critical care parameters such as Procalcitonin, Troponin I, CK-MB, Myoglobin, NT-proBNP, and D-Dimer as well as tests for sphingotec’s proprietary biomarkers such as DPP3, an assay for Dipeptidyl Peptidase 3, a unique and proprietary biomarker for cardio-renal pathway disruptions leading to acute organ dysfunction, and Proenkephalin (penKid®), a unique and proprietary biomarker for real-time assessment of kidney function. An IB10 assay for bioactive Adrenomedullin (bio-ADM®), a unique and proprietary biomarker for endothelial function is expected to be launched later in 2020.
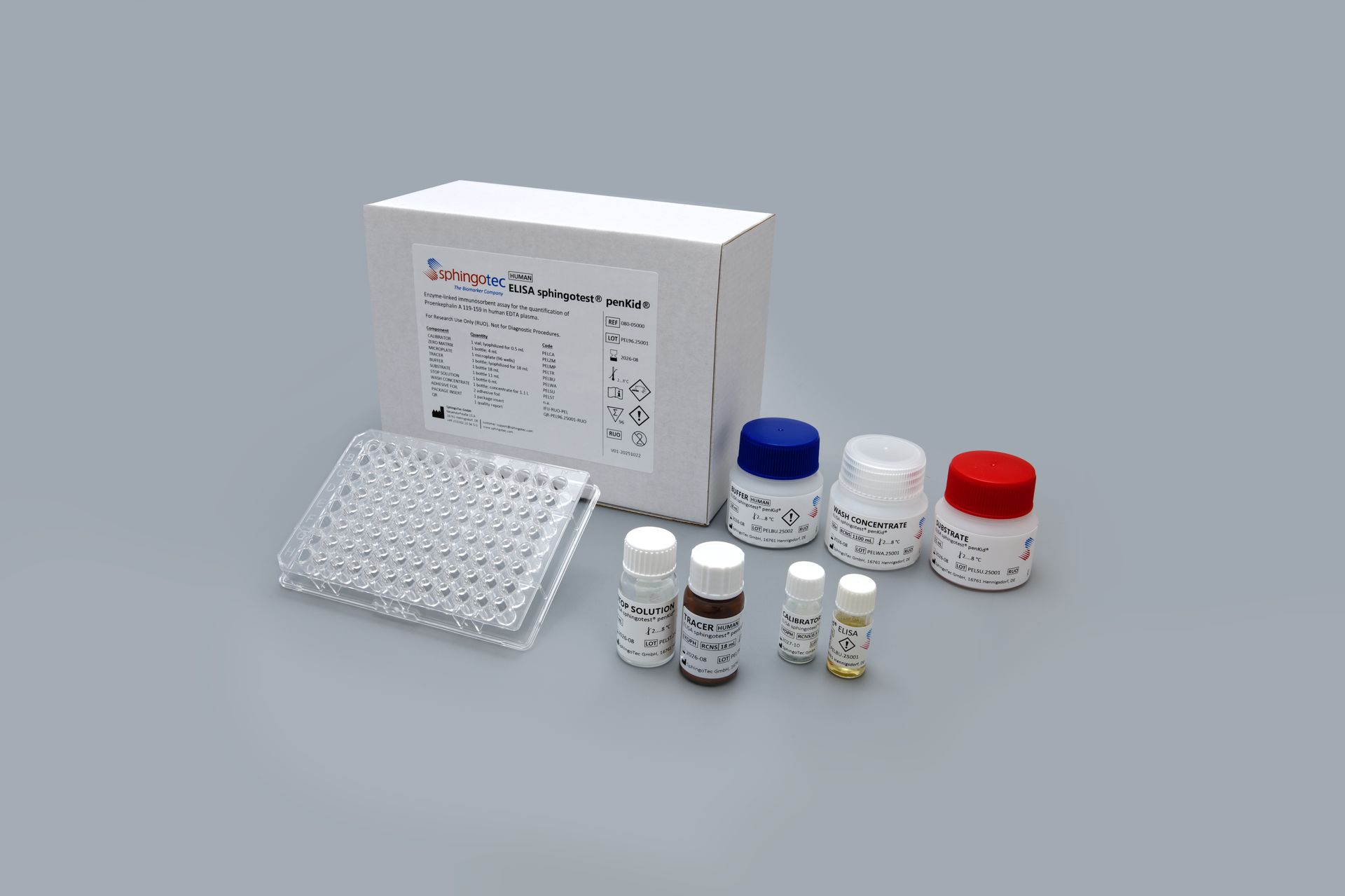
• New ELISA sphingotest® penKid® enables widespread measurement of the kidney function biomarker Proenkephalin 119-159 (penKid) using standard laboratory equipment. • Only available test to match SphingoTec’s reference chemiluminescence assay, developed with patented high-sensitivity technology to provide consistent results across platforms. Hennigsdorf/Berlin, Germany, April 28 2026 - SphingoTec GmbH announces the launch of the ELISA sphingotest® penKid®, a new assay designed to make testing of its proprietary biomarker broadly accessible to research laboratories and pharmaceutical partners. Providing ease-of-use and precision, the test facilitates large-scale investigations of human kidney function in both acute and broader clinical research contexts. Responding to growing research interest The launch follows increasing demand from the scientific community to study penKid - a biomarker reflecting the current state of kidney function in critical care environments. While SphingoTec’s high-sensitivity sphingotest® penKid® assay serves as the reference method for clinical research and third-party IVD assays, the assay technology requiring dedicated equipment was not easily adoptable in research laboratories. The new ELISA sphingotest® penKid® now allows researchers worldwide to benefit from SphingoTec’s patented detection technology using standard photometers, extending penKid testing capability to a wider range of laboratories. Proven performance and data continuity The ELISA sphingotest® penKid® has been developed to achieve excellent correlation with SphingoTec’s reference chemiluminescence assay, ensuring consistent, high-quality results across different assay platforms. Both methods share the same underlying technology capable of detecting penKid concentrations in the picomolar range. Thanks to this technology transfer, researchers can rely on the same analytical precision and reliability that supported the clinical studies establishing penKid as a valuable kidney function biomarker. The assay’s robust design and German manufacturing ensure durable consistency and reproducibility. With this research-use ELISA, SphingoTec responds to the increasing availability of non-validated assays and encourages researchers to not compromise assay quality and performance to ensure trustworthy results - since data from non-validated tests may reflect assay limitations rather than the true performance of the penKid biomarker. Complementary approaches for clinical use and scientific exploration SphingoTec pursues its commercial strategy for the biomarker penKid through strategic out-licensing partnerships such as Boditech Med, which has developed an IVDR-certified assay for routine clinical use and rapid diagnostics. The newly launched ELISA sphingotest® penKid® represents a complementary initiative, specifically designed to support high-throughput clinical and translational research. Through this dual approach, SphingoTec reaffirms its commitment to fostering scientific collaboration and promoting the broader exploration of penKid across diverse research settings. “Developing the ELISA sphingotest® penKid® reflects our long-term commitment to improving critical care diagnostics,” said Deborah Bergmann, Managing Director and CEO of SphingoTec GmbH. “Beyond its currently validated clinical applications, we see strong potential for penKid to support research and improve diagnostics in other fields where kidney function is relevant. By encouraging scientists worldwide to incorporate penKid into their studies and clinical programs, we aim to accelerate innovation and advance best-fit diagnostic solutions that ultimately improve patient care.” About SphingoTec SphingoTec GmbH ("SphingoTec"; Hennigsdorf near Berlin, Germany) is a biomarker company focusing on the out-licensing of innovative critical care solutions for diagnosing, predicting, and monitoring acute medical conditions. SphingoTec develops its biomarkers to the commercial stage and partners with IVD companies to make them available on different IVD platforms. SphingoTec's proprietary biomarker portfolio includes Proenkephalin A 119-159 (penKid), a biomarker for the assessment of kidney function in critical diseases, and bioactive Adrenomedullin 1-52 (bio-ADM), a biomarker for the assessment of endothelial function in conditions like sepsis. Discover more on www.sphingotec.com Media Contact: Email: press@sphingotec.com Phone +49-3302-20565-0 SphingoTec GmbH Neuendorfstr. 15A 16761 Hennigsdorf, Germany
• PenKid surpasses serum creatinine on Day 1 post-transplant in detecting delayed graft function (DGF), with an AUROC of 0.87 versus 0.56 for creatinine. • PenKid differentiates slow graft function (SGF) from DGF up to 8 days earlier than current methods, supporting more timely clinical decisions. • PenKid levels remain unaffected by kidney replacement therapy (KRT), allowing for more accurate assessment of kidney function. • Independent validation in transplant cohort from Australia confirms performance and broad applicability. Hennigsdorf/Berlin, Germany, July 1, 2025 - Diagnostic company SphingoTec GmbH (“SphingoTec”) announces a landmark study (1) published in Transplant International, led by Heidelberg University Hospital in Germany in collaboration with researchers from Sydney, Australia, which identifies Proenkephalin A 119-159 (penKid) as a reliable biomarker for early and precise assessment of graft function trajectories following kidney transplantation. The research demonstrates that PenKid not only identifies patients at risk for DGF significantly earlier than traditional markers but also distinguishes between slow and delayed graft function with remarkable accuracy, offering clinicians a valuable new tool for patient management. The study prospectively evaluated 159 consecutive kidney transplant recipients at Heidelberg University Hospital and validated findings in an independent cohort from Sydney. PenKid consistently outperformed serum creatinine (SCr) in predicting graft function trajectories, particularly in the critical early post-transplant period. Notably, PenKid’s ability to remain unaffected by KRT—a treatment for severe kidney dysfunction—further sets it apart from SCr, which can be influenced by non-renal factors and KRT itself, thereby enhancing the reliability of graft function assessment. Multivariate analysis confirmed PenKid as the strongest independent predictor of both short-term graft function and 30-day outcomes, underscoring its clinical utility for early risk stratification. The biomarker’s superior granularity allows for nuanced classification of DGF severity, supporting more informed decisions regarding the initiation of dialysis or biopsy and offering potential for individualized patient care. With these findings, penKid steps forward as a practical addition to the transplant clinician’s toolkit, promising to sharpen decision-making for optimal outcomes. Its adoption could help transplant teams act with greater confidence and precision, ultimately strengthening the standard of care in kidney transplantation. ## References 1. Benning L et al. (2025) Proenkephalin A 119-159 in Kidney Transplantation: A Novel Biomarker for Superior Tracking of Graft Function Trajectories. Transpl. Int. 38:14366. doi: 10.3389/ti.2025.14366 About SphingoTec SphingoTec GmbH ("SphingoTec"; Hennigsdorf near Berlin, Germany) is a biomarker company focusing on the out-licensing of innovative critical care solutions for diagnosing, predicting, and monitoring acute medical conditions. SphingoTec develops its biomarkers to the commercial stage and partners with IVD companies to make them available on different IVD platforms. SphingoTec's proprietary biomarker portfolio includes Proenkephalin A 119-159 (penKid), a biomarker for the assessment of kidney function in critical diseases, commercially available on diagnostic platforms AFIAS and Nexus IB10 and bioactive Adrenomedullin 1-52 (bio-ADM), a biomarker for the assessment of endothelial function in conditions like sepsis. Discover more on www.sphingotec.com Contact : Ruxandra Lenz Marketing and Communication SphingoTec GmbH Phone +49-3302-20565-0 Email: press@sphingotec.com
