Majority of Women Do Not Discuss Breast Cancer Risks with Healthcare Provider
Cambridge, Mass. – May 5, 2015 sphingotec, LLC, a developer of predictive diagnostic tests for cancer, cardiovascular diseases and kidney function, today announced the results of a survey gauging women’s understanding of breast cancer risk factors and their perceptions of common breast cancer screening methods.
The survey was conducted in conjunction with a national, independent health information resource for women, and found that of the 464 anonymous respondents, the majority of women do not discuss breast cancer risk factors or the guidelines and limitations of mammography screenings with their healthcare providers.
“Mammograms remain an important screening tool and can save lives by helping detect some cases of breast cancer early, however women are not aware of the limitations of mammography, nor are they aware of their personal risk factors leading to breast cancer, according to the survey,” said Karla Gonye, president, sphingotec, LLC. “The results of the survey give us insight into the market need for breast cancer biomarkers beyond BRCA 1/2 and further indicate the desire for more risk predictive tests so that women and their doctors can make more informed decisions about their risk of breast cancer.”
The top findings from the survey include:
Overwhelmingly, women want to know more about their personal risk for breast cancer, but rarely or never discuss their risk factors with a healthcare provider.
Nearly all women (99 percent) expressed interest in understanding their personal risk for breast cancer.
However, 65 percent rarely or never discuss their breast cancer risk factors with a healthcare provider, a concerning statistic given that breast cancer is the most common cancer among women.
Those surveyed identified family history (94 percent), use of hormone replacement therapy (73 percent), smoking (64 percent) and age (58 percent) as the top risk factors for breast cancer.
Despite conflicting recommendations around the frequency of mammogram screening and growing evidence that annual screenings can lead to added expense and anxiety due to false-positive results, the majority of women still get annual mammograms.
Sixty-four percent of women get a mammogram once a year.
Nineteen percent get a mammogram every two to three years.
Given that mammograms miss one in five occurrences of breast cancer1, mammography screenings need to be complemented with new and more affordable prognostic tools for assessing and predicting breast cancer risk.
Upon receiving a normal mammogram result, more than half of women (55 percent) believed that they were cancer free, but only 45 percent were aware of the limitations of mammograms in the early detection of breast cancer.
Ninety-six percent of women indicated they want a simple and affordable blood test to determine breast cancer risk.
1National Cancer Institute, https://www.cancer.gov/cancertopics/types/breast/mammograms-fact-sheet
Survey Methodology
This independent survey was conducted online through SurveyMonkey on behalf of sphingotec from April 20 – 23, 2015 among 464 anonymous women living in the United States. Ninety percent of the women surveyed were over the age of 40 and 10 percent were between 20 – 39 years old.
About sphingotec
The biotechnology company SphingoTec GmbH, headquartered in Hennigsdorf, Germany with U.S. operations in Cambridge, Massachusetts, was founded in 2002. sphingotec develops diagnostic methods for prediction, prevention, intervention strategies and early treatment of diseases in the fields of cancer, cardiovascular diseases and kidney function. Using biomarkers to indicate susceptibility for a specific disease provides individuals with knowledge that they are at risk. The biomarkers therefore create a starting point where intervention strategies can be employed, as evidence-based recommendations on how to reduce risks are always an integral part of the complete concept. The company’s sphingotest pro-NT and sphingotest pro-ENK tests take a unique approach to breast cancer prediction that is unlike any other risk assessment test currently available. The tests identify the concentration of the peptides neurotensin (NT) and enkaphalin (ENK) in the blood, which can then determine individual risk levels. Further information can be found on our website www.sphingotec.com.
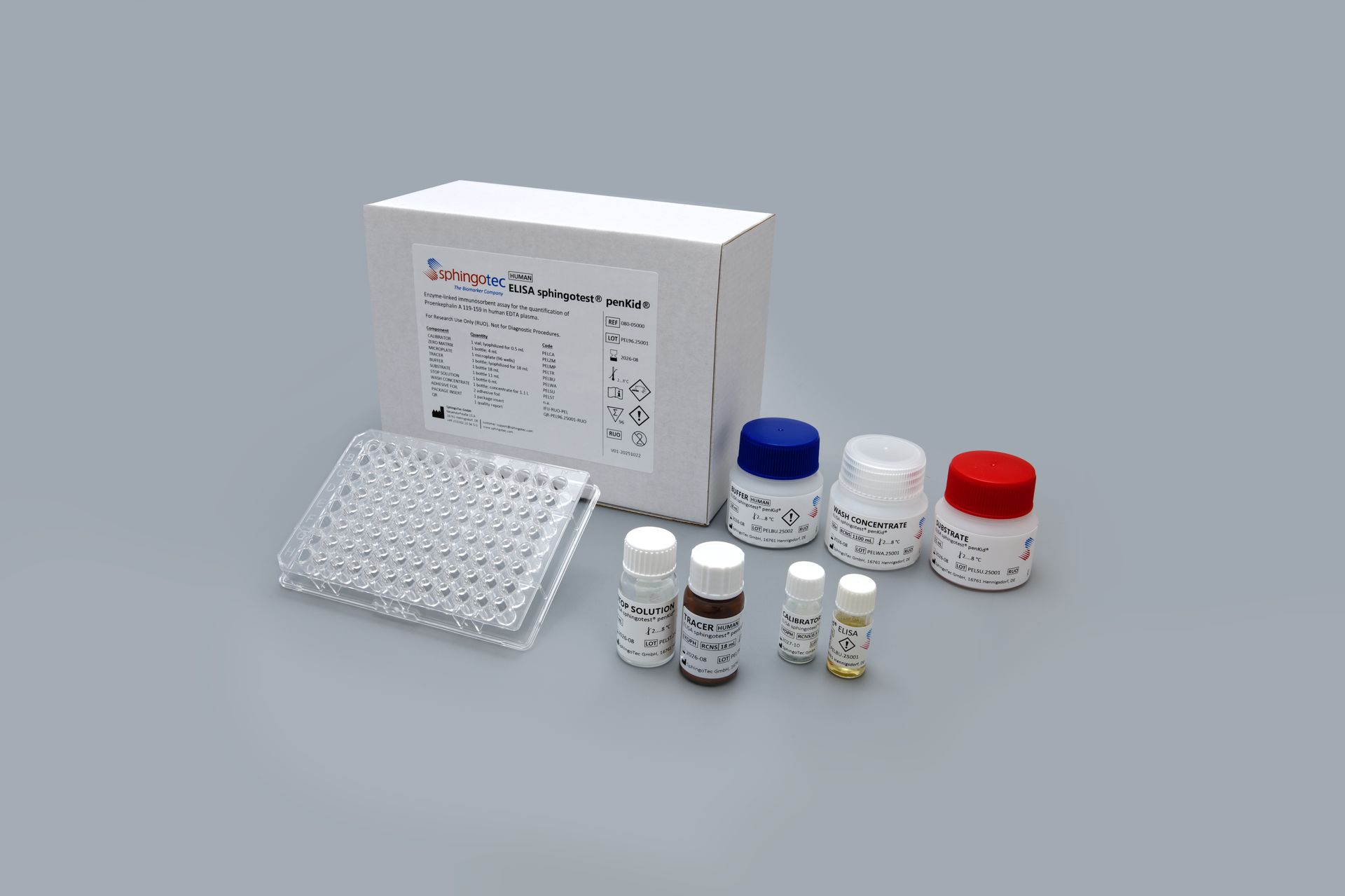
• New ELISA sphingotest® penKid® enables widespread measurement of the kidney function biomarker Proenkephalin 119-159 (penKid) using standard laboratory equipment. • Only available test to match SphingoTec’s reference chemiluminescence assay, developed with patented high-sensitivity technology to provide consistent results across platforms. Hennigsdorf/Berlin, Germany, April 28 2026 - SphingoTec GmbH announces the launch of the ELISA sphingotest® penKid®, a new assay designed to make testing of its proprietary biomarker broadly accessible to research laboratories and pharmaceutical partners. Providing ease-of-use and precision, the test facilitates large-scale investigations of human kidney function in both acute and broader clinical research contexts. Responding to growing research interest The launch follows increasing demand from the scientific community to study penKid - a biomarker reflecting the current state of kidney function in critical care environments. While SphingoTec’s high-sensitivity sphingotest® penKid® assay serves as the reference method for clinical research and third-party IVD assays, the assay technology requiring dedicated equipment was not easily adoptable in research laboratories. The new ELISA sphingotest® penKid® now allows researchers worldwide to benefit from SphingoTec’s patented detection technology using standard photometers, extending penKid testing capability to a wider range of laboratories. Proven performance and data continuity The ELISA sphingotest® penKid® has been developed to achieve excellent correlation with SphingoTec’s reference chemiluminescence assay, ensuring consistent, high-quality results across different assay platforms. Both methods share the same underlying technology capable of detecting penKid concentrations in the picomolar range. Thanks to this technology transfer, researchers can rely on the same analytical precision and reliability that supported the clinical studies establishing penKid as a valuable kidney function biomarker. The assay’s robust design and German manufacturing ensure durable consistency and reproducibility. With this research-use ELISA, SphingoTec responds to the increasing availability of non-validated assays and encourages researchers to not compromise assay quality and performance to ensure trustworthy results - since data from non-validated tests may reflect assay limitations rather than the true performance of the penKid biomarker. Complementary approaches for clinical use and scientific exploration SphingoTec pursues its commercial strategy for the biomarker penKid through strategic out-licensing partnerships such as Boditech Med, which has developed an IVDR-certified assay for routine clinical use and rapid diagnostics. The newly launched ELISA sphingotest® penKid® represents a complementary initiative, specifically designed to support high-throughput clinical and translational research. Through this dual approach, SphingoTec reaffirms its commitment to fostering scientific collaboration and promoting the broader exploration of penKid across diverse research settings. “Developing the ELISA sphingotest® penKid® reflects our long-term commitment to improving critical care diagnostics,” said Deborah Bergmann, Managing Director and CEO of SphingoTec GmbH. “Beyond its currently validated clinical applications, we see strong potential for penKid to support research and improve diagnostics in other fields where kidney function is relevant. By encouraging scientists worldwide to incorporate penKid into their studies and clinical programs, we aim to accelerate innovation and advance best-fit diagnostic solutions that ultimately improve patient care.” About SphingoTec SphingoTec GmbH ("SphingoTec"; Hennigsdorf near Berlin, Germany) is a biomarker company focusing on the out-licensing of innovative critical care solutions for diagnosing, predicting, and monitoring acute medical conditions. SphingoTec develops its biomarkers to the commercial stage and partners with IVD companies to make them available on different IVD platforms. SphingoTec's proprietary biomarker portfolio includes Proenkephalin A 119-159 (penKid), a biomarker for the assessment of kidney function in critical diseases, and bioactive Adrenomedullin 1-52 (bio-ADM), a biomarker for the assessment of endothelial function in conditions like sepsis. Discover more on www.sphingotec.com Media Contact: Email: press@sphingotec.com Phone +49-3302-20565-0 SphingoTec GmbH Neuendorfstr. 15A 16761 Hennigsdorf, Germany
• PenKid surpasses serum creatinine on Day 1 post-transplant in detecting delayed graft function (DGF), with an AUROC of 0.87 versus 0.56 for creatinine. • PenKid differentiates slow graft function (SGF) from DGF up to 8 days earlier than current methods, supporting more timely clinical decisions. • PenKid levels remain unaffected by kidney replacement therapy (KRT), allowing for more accurate assessment of kidney function. • Independent validation in transplant cohort from Australia confirms performance and broad applicability. Hennigsdorf/Berlin, Germany, July 1, 2025 - Diagnostic company SphingoTec GmbH (“SphingoTec”) announces a landmark study (1) published in Transplant International, led by Heidelberg University Hospital in Germany in collaboration with researchers from Sydney, Australia, which identifies Proenkephalin A 119-159 (penKid) as a reliable biomarker for early and precise assessment of graft function trajectories following kidney transplantation. The research demonstrates that PenKid not only identifies patients at risk for DGF significantly earlier than traditional markers but also distinguishes between slow and delayed graft function with remarkable accuracy, offering clinicians a valuable new tool for patient management. The study prospectively evaluated 159 consecutive kidney transplant recipients at Heidelberg University Hospital and validated findings in an independent cohort from Sydney. PenKid consistently outperformed serum creatinine (SCr) in predicting graft function trajectories, particularly in the critical early post-transplant period. Notably, PenKid’s ability to remain unaffected by KRT—a treatment for severe kidney dysfunction—further sets it apart from SCr, which can be influenced by non-renal factors and KRT itself, thereby enhancing the reliability of graft function assessment. Multivariate analysis confirmed PenKid as the strongest independent predictor of both short-term graft function and 30-day outcomes, underscoring its clinical utility for early risk stratification. The biomarker’s superior granularity allows for nuanced classification of DGF severity, supporting more informed decisions regarding the initiation of dialysis or biopsy and offering potential for individualized patient care. With these findings, penKid steps forward as a practical addition to the transplant clinician’s toolkit, promising to sharpen decision-making for optimal outcomes. Its adoption could help transplant teams act with greater confidence and precision, ultimately strengthening the standard of care in kidney transplantation. ## References 1. Benning L et al. (2025) Proenkephalin A 119-159 in Kidney Transplantation: A Novel Biomarker for Superior Tracking of Graft Function Trajectories. Transpl. Int. 38:14366. doi: 10.3389/ti.2025.14366 About SphingoTec SphingoTec GmbH ("SphingoTec"; Hennigsdorf near Berlin, Germany) is a biomarker company focusing on the out-licensing of innovative critical care solutions for diagnosing, predicting, and monitoring acute medical conditions. SphingoTec develops its biomarkers to the commercial stage and partners with IVD companies to make them available on different IVD platforms. SphingoTec's proprietary biomarker portfolio includes Proenkephalin A 119-159 (penKid), a biomarker for the assessment of kidney function in critical diseases, commercially available on diagnostic platforms AFIAS and Nexus IB10 and bioactive Adrenomedullin 1-52 (bio-ADM), a biomarker for the assessment of endothelial function in conditions like sepsis. Discover more on www.sphingotec.com Contact : Ruxandra Lenz Marketing and Communication SphingoTec GmbH Phone +49-3302-20565-0 Email: press@sphingotec.com
