sphingotec at the St. Gallen International Breast Cancer Conference
Cambridge, Mass. – Feb. 4, 2015 –
sphingotec LLC announced today that the findings from its research seeking to improve breast cancer risk stratification for guiding hormone replacement therapy has been selected for presentation at the 14th annual St. Gallen International Breast Cancer Conference in Vienna, Austria, March 18 – 21, 2015. The poster will be presented by Dr. Uwe Hantke.
sphingotec’s study assessed how plasma pro-neurotensin (pro-NT) and pro-enkephalin (pro-ENK) help predict whether women have high or low risk for developing breast cancer so physicians can make a more informed decision on whether to use hormone replacement therapy. The results concluded that both plasma pro-NT and pro-ENK provide new and independent information above and beyond commonly known breast cancer risk factors that can improve risk stratification.
As further evidence of the power of pro-NT and pro-ENK risk prediction, a theoretical model demonstrated that 10 out of 123 breast cancer occurrences could have potentially been avoided if clinicians knew their patients’ pro-NT and pro-ENK levels put them in an extremely high-risk group and used this information to avoid HRT.
Although extensive research has been conducted around the risks and benefits of HRT there is disagreement on whether the benefits outweigh the risks. According to The Centers for Disease Control and Prevention (CDC), those who caution against using HRT often refer to the increased risk of endometrial and breast cancer associated with using estrogen, especially for extended lengths of time. Those who advocate for its use often point out that HRT appears to reduce the risk of osteoporosis, heart disease and Alzheimer’s disease in addition to alleviating menopausal symptoms. The controversy surrounding HRT leaves clinicians and their patients feeling uncertain about individual breast cancer risk, one of the most common types of cancer for women.
“Studies have shown that hormone replacement therapy can increase the risk of breast cancer, but it varies greatly based on each woman’s individual characteristics and risk factors,” said Dr. Andreas Bergmann, CEO of SphingoTec GmbH. “There is a huge unmet need for improved risk stratification to help doctors decide whether to recommend hormone replacement therapy. We believe our research will enable doctors and their patients to make a more educated decision on whether this treatment is right for them.”
To conduct the study, sphingotec researchers measured pro-ENK and pro-NT in fasting plasma from 1,929 women participating in the Malmö Diet and Cancer Study and used Cox proportional hazards models to relate pro-ENK and pro-NT to first breast cancer events within 15 years of follow up. For the cohort design, they sampled 1,569 women from the Malmö Preventive Project and used multivariate adjusted logistic regression models to relate pro-ENK and pro-NT to first breast cancer events during the observation period. They then analyzed subgroups of women who received HRT to assess the performance of each marker.
This study supports sphingotec’s mission to reduce the incidence of major illnesses such as cancer, cardiovascular conditions and kidney disease through tests that can detect individual risk factors. sphingotec has developed two simple blood tests, sphingotest® pro-NT and the sphingotest® pro-ENK that identifies the concentration of these peptides in the blood. The biomarkers are independently associated with the development of breast cancer above other commonly known risk factors and are applicable to all females, regardless of genetic predispositions. Both pro-neurotensin and pro-enkephalin assays will be available to the U.S. and European markets later this year.
sphingotec also announced today that the breast cancer biomarkers pro-NT and pro-ENK will be featured in a mid-morning session at the American Association of Clinical Chemistry (AACC) Annual Meeting & Clinical Lab Expo in Atlanta, Georgia July 26 – 30, 2015. A panel of leading researchers will address the gaps and limitations in breast cancer prevention programs and how emerging biomarkers can help clinicians and laboratorians address the key challenges. The panelists are:
Dr. Max Wicha, distinguished professor of oncology and director of the Comprehensive Cancer Center at University of Michigan
Dr. Allan Jaffe, M.D., Mayo Clinic cardiologist and division chair, Clinical Core Laboratory Services
Dr. Alan Maisel, director of the Coronary Care Unit and Heart Failure Program at the VA San Diego Healthcare System, Professor of medicine at University of California, San Diego.
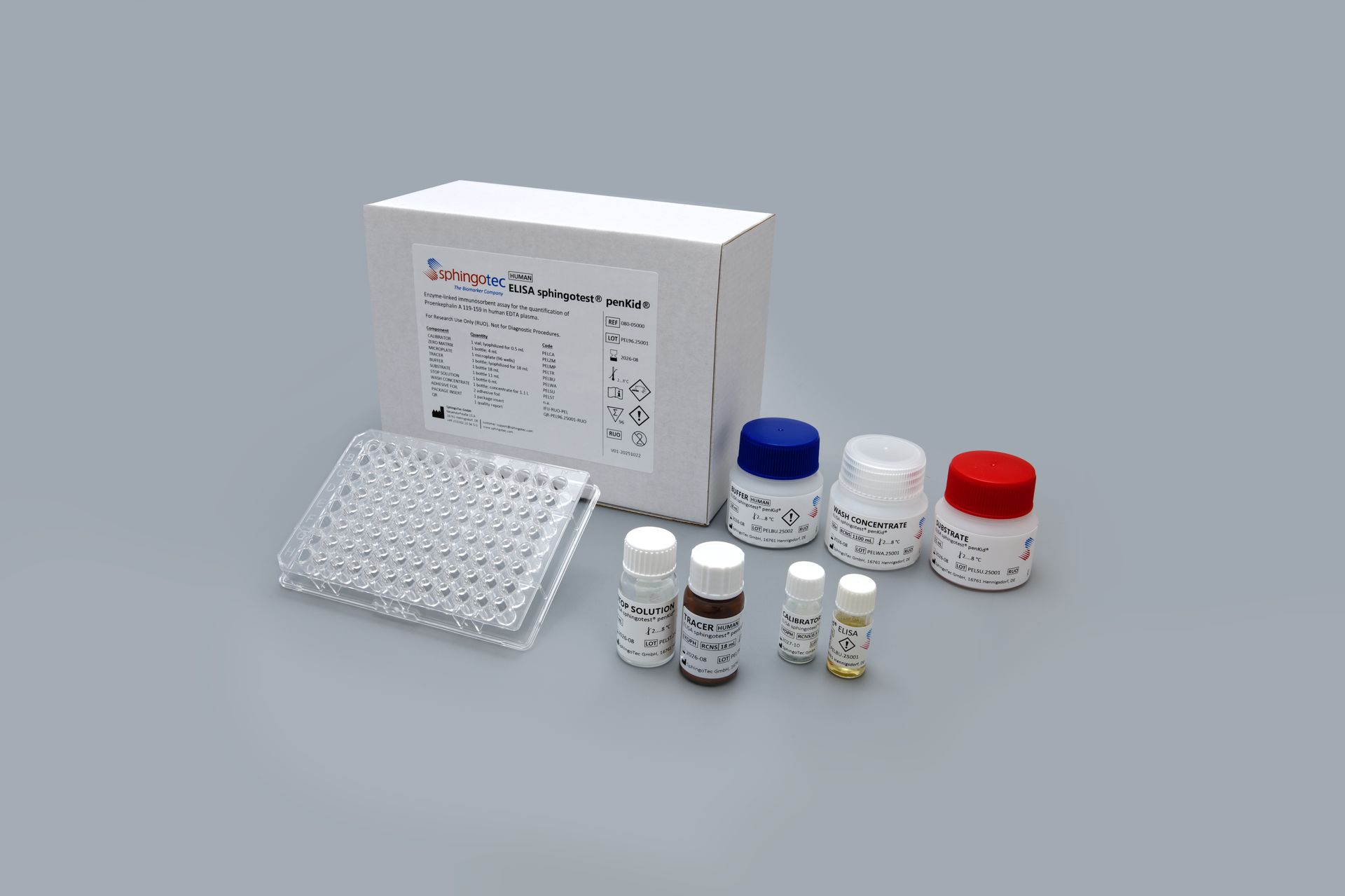
• New ELISA sphingotest® penKid® enables widespread measurement of the kidney function biomarker Proenkephalin 119-159 (penKid) using standard laboratory equipment. • Only available test to match SphingoTec’s reference chemiluminescence assay, developed with patented high-sensitivity technology to provide consistent results across platforms. Hennigsdorf/Berlin, Germany, April 28 2026 - SphingoTec GmbH announces the launch of the ELISA sphingotest® penKid®, a new assay designed to make testing of its proprietary biomarker broadly accessible to research laboratories and pharmaceutical partners. Providing ease-of-use and precision, the test facilitates large-scale investigations of human kidney function in both acute and broader clinical research contexts. Responding to growing research interest The launch follows increasing demand from the scientific community to study penKid - a biomarker reflecting the current state of kidney function in critical care environments. While SphingoTec’s high-sensitivity sphingotest® penKid® assay serves as the reference method for clinical research and third-party IVD assays, the assay technology requiring dedicated equipment was not easily adoptable in research laboratories. The new ELISA sphingotest® penKid® now allows researchers worldwide to benefit from SphingoTec’s patented detection technology using standard photometers, extending penKid testing capability to a wider range of laboratories. Proven performance and data continuity The ELISA sphingotest® penKid® has been developed to achieve excellent correlation with SphingoTec’s reference chemiluminescence assay, ensuring consistent, high-quality results across different assay platforms. Both methods share the same underlying technology capable of detecting penKid concentrations in the picomolar range. Thanks to this technology transfer, researchers can rely on the same analytical precision and reliability that supported the clinical studies establishing penKid as a valuable kidney function biomarker. The assay’s robust design and German manufacturing ensure durable consistency and reproducibility. With this research-use ELISA, SphingoTec responds to the increasing availability of non-validated assays and encourages researchers to not compromise assay quality and performance to ensure trustworthy results - since data from non-validated tests may reflect assay limitations rather than the true performance of the penKid biomarker. Complementary approaches for clinical use and scientific exploration SphingoTec pursues its commercial strategy for the biomarker penKid through strategic out-licensing partnerships such as Boditech Med, which has developed an IVDR-certified assay for routine clinical use and rapid diagnostics. The newly launched ELISA sphingotest® penKid® represents a complementary initiative, specifically designed to support high-throughput clinical and translational research. Through this dual approach, SphingoTec reaffirms its commitment to fostering scientific collaboration and promoting the broader exploration of penKid across diverse research settings. “Developing the ELISA sphingotest® penKid® reflects our long-term commitment to improving critical care diagnostics,” said Deborah Bergmann, Managing Director and CEO of SphingoTec GmbH. “Beyond its currently validated clinical applications, we see strong potential for penKid to support research and improve diagnostics in other fields where kidney function is relevant. By encouraging scientists worldwide to incorporate penKid into their studies and clinical programs, we aim to accelerate innovation and advance best-fit diagnostic solutions that ultimately improve patient care.” About SphingoTec SphingoTec GmbH ("SphingoTec"; Hennigsdorf near Berlin, Germany) is a biomarker company focusing on the out-licensing of innovative critical care solutions for diagnosing, predicting, and monitoring acute medical conditions. SphingoTec develops its biomarkers to the commercial stage and partners with IVD companies to make them available on different IVD platforms. SphingoTec's proprietary biomarker portfolio includes Proenkephalin A 119-159 (penKid), a biomarker for the assessment of kidney function in critical diseases, and bioactive Adrenomedullin 1-52 (bio-ADM), a biomarker for the assessment of endothelial function in conditions like sepsis. Discover more on www.sphingotec.com Media Contact: Email: press@sphingotec.com Phone +49-3302-20565-0 SphingoTec GmbH Neuendorfstr. 15A 16761 Hennigsdorf, Germany
• PenKid surpasses serum creatinine on Day 1 post-transplant in detecting delayed graft function (DGF), with an AUROC of 0.87 versus 0.56 for creatinine. • PenKid differentiates slow graft function (SGF) from DGF up to 8 days earlier than current methods, supporting more timely clinical decisions. • PenKid levels remain unaffected by kidney replacement therapy (KRT), allowing for more accurate assessment of kidney function. • Independent validation in transplant cohort from Australia confirms performance and broad applicability. Hennigsdorf/Berlin, Germany, July 1, 2025 - Diagnostic company SphingoTec GmbH (“SphingoTec”) announces a landmark study (1) published in Transplant International, led by Heidelberg University Hospital in Germany in collaboration with researchers from Sydney, Australia, which identifies Proenkephalin A 119-159 (penKid) as a reliable biomarker for early and precise assessment of graft function trajectories following kidney transplantation. The research demonstrates that PenKid not only identifies patients at risk for DGF significantly earlier than traditional markers but also distinguishes between slow and delayed graft function with remarkable accuracy, offering clinicians a valuable new tool for patient management. The study prospectively evaluated 159 consecutive kidney transplant recipients at Heidelberg University Hospital and validated findings in an independent cohort from Sydney. PenKid consistently outperformed serum creatinine (SCr) in predicting graft function trajectories, particularly in the critical early post-transplant period. Notably, PenKid’s ability to remain unaffected by KRT—a treatment for severe kidney dysfunction—further sets it apart from SCr, which can be influenced by non-renal factors and KRT itself, thereby enhancing the reliability of graft function assessment. Multivariate analysis confirmed PenKid as the strongest independent predictor of both short-term graft function and 30-day outcomes, underscoring its clinical utility for early risk stratification. The biomarker’s superior granularity allows for nuanced classification of DGF severity, supporting more informed decisions regarding the initiation of dialysis or biopsy and offering potential for individualized patient care. With these findings, penKid steps forward as a practical addition to the transplant clinician’s toolkit, promising to sharpen decision-making for optimal outcomes. Its adoption could help transplant teams act with greater confidence and precision, ultimately strengthening the standard of care in kidney transplantation. ## References 1. Benning L et al. (2025) Proenkephalin A 119-159 in Kidney Transplantation: A Novel Biomarker for Superior Tracking of Graft Function Trajectories. Transpl. Int. 38:14366. doi: 10.3389/ti.2025.14366 About SphingoTec SphingoTec GmbH ("SphingoTec"; Hennigsdorf near Berlin, Germany) is a biomarker company focusing on the out-licensing of innovative critical care solutions for diagnosing, predicting, and monitoring acute medical conditions. SphingoTec develops its biomarkers to the commercial stage and partners with IVD companies to make them available on different IVD platforms. SphingoTec's proprietary biomarker portfolio includes Proenkephalin A 119-159 (penKid), a biomarker for the assessment of kidney function in critical diseases, commercially available on diagnostic platforms AFIAS and Nexus IB10 and bioactive Adrenomedullin 1-52 (bio-ADM), a biomarker for the assessment of endothelial function in conditions like sepsis. Discover more on www.sphingotec.com Contact : Ruxandra Lenz Marketing and Communication SphingoTec GmbH Phone +49-3302-20565-0 Email: press@sphingotec.com
